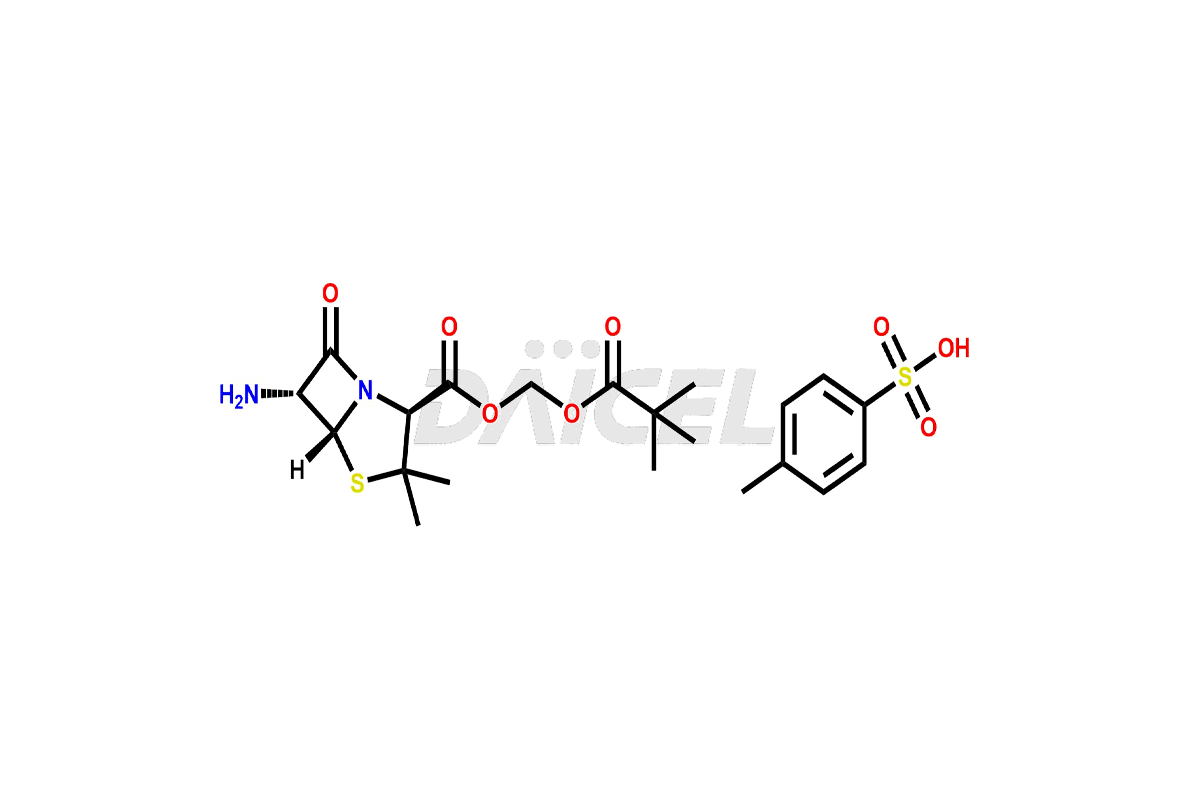
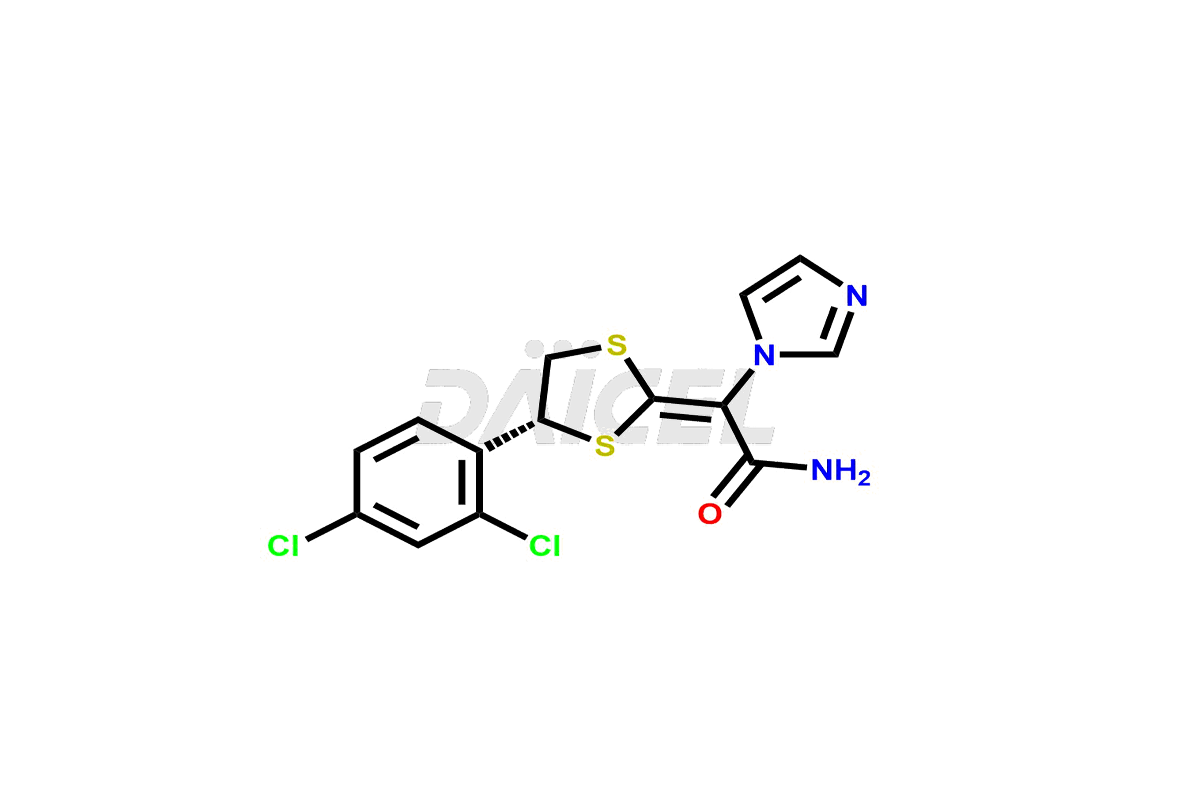
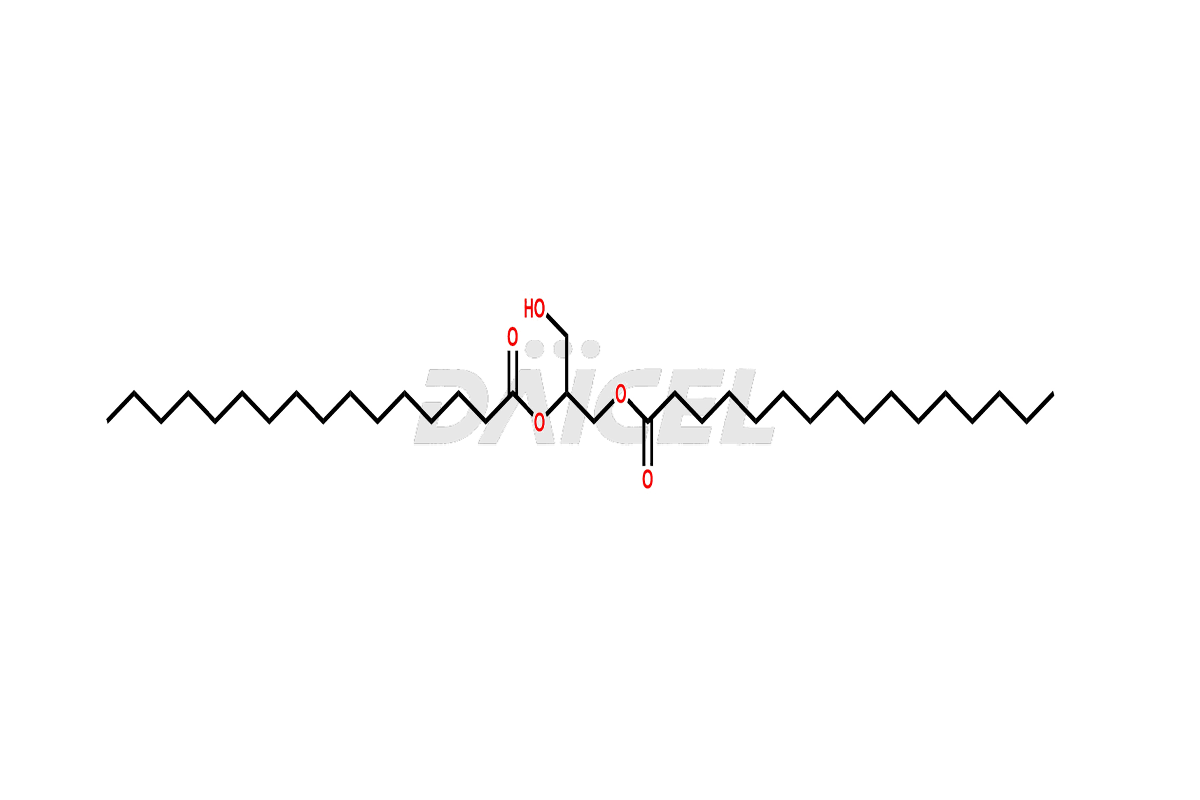
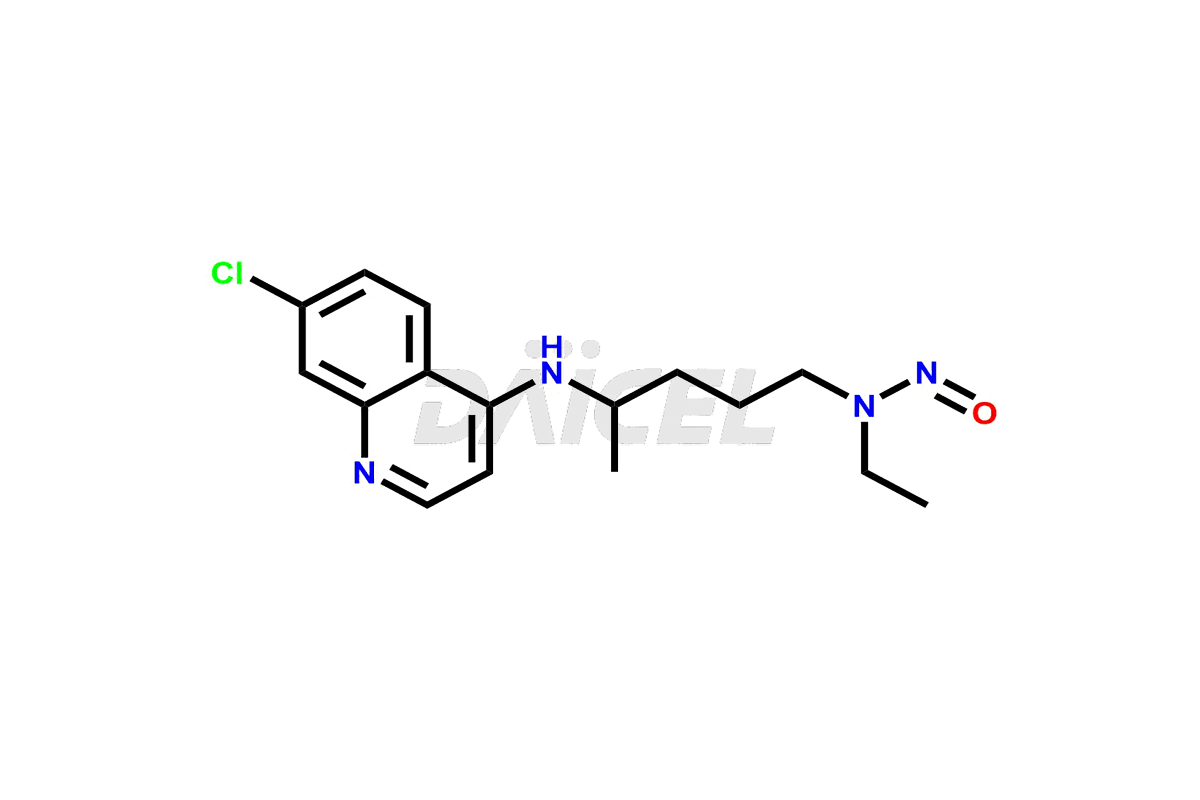
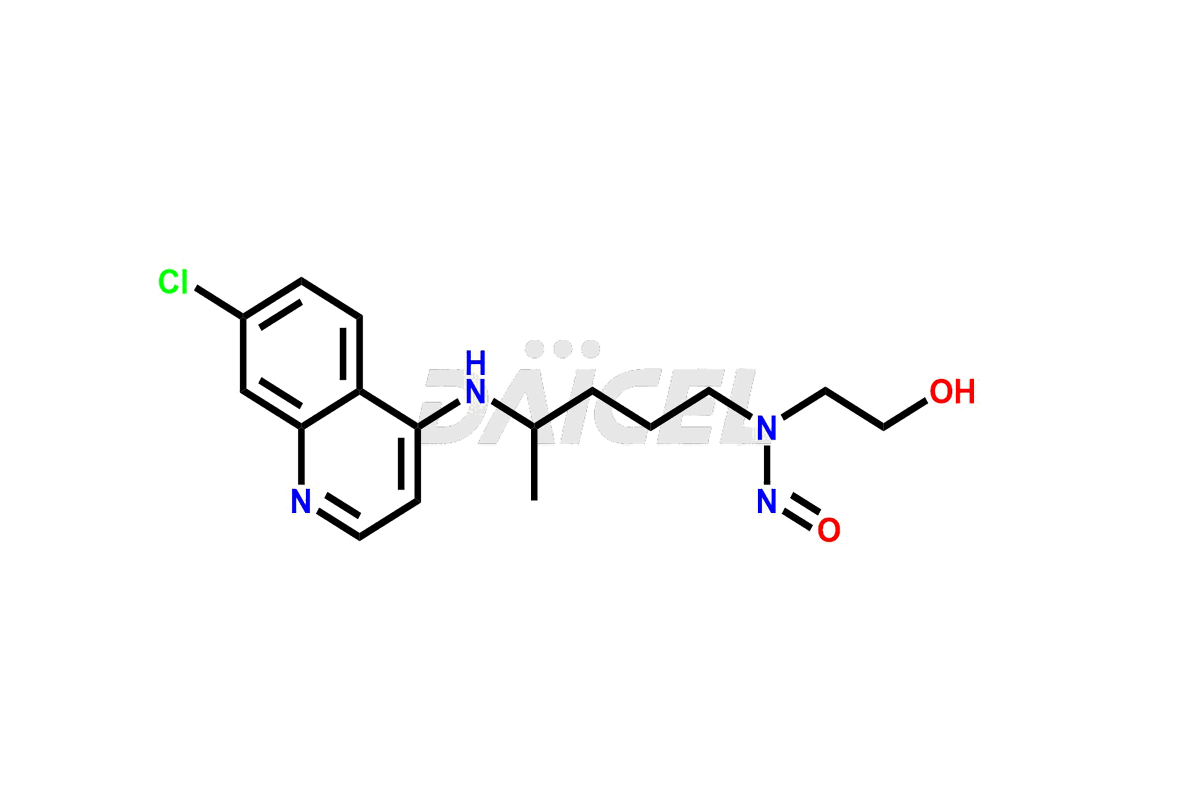
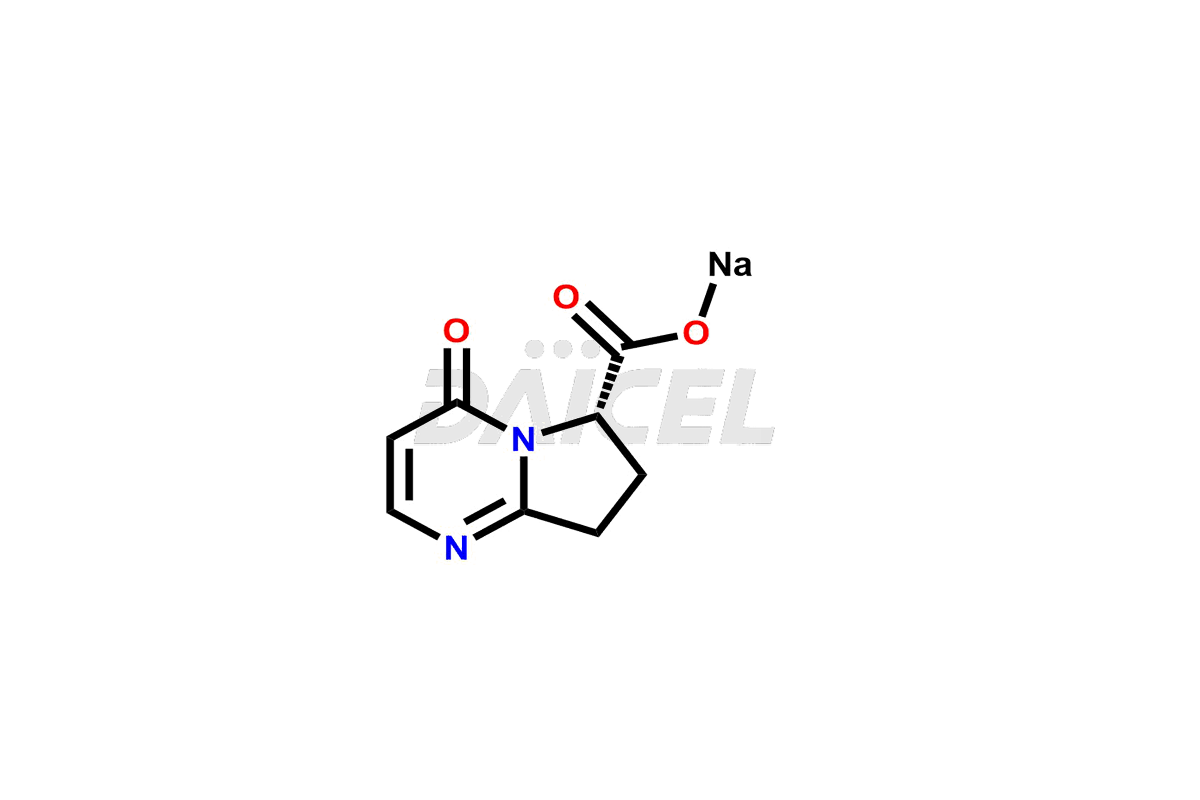
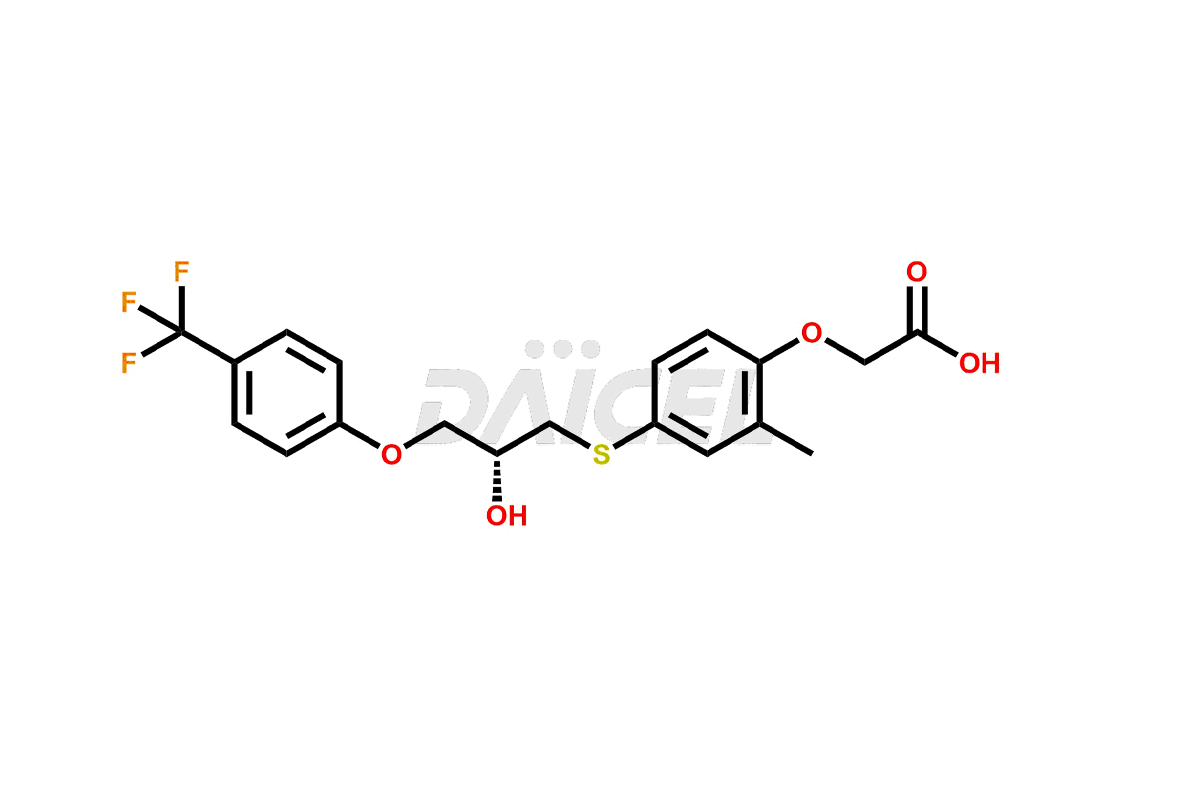
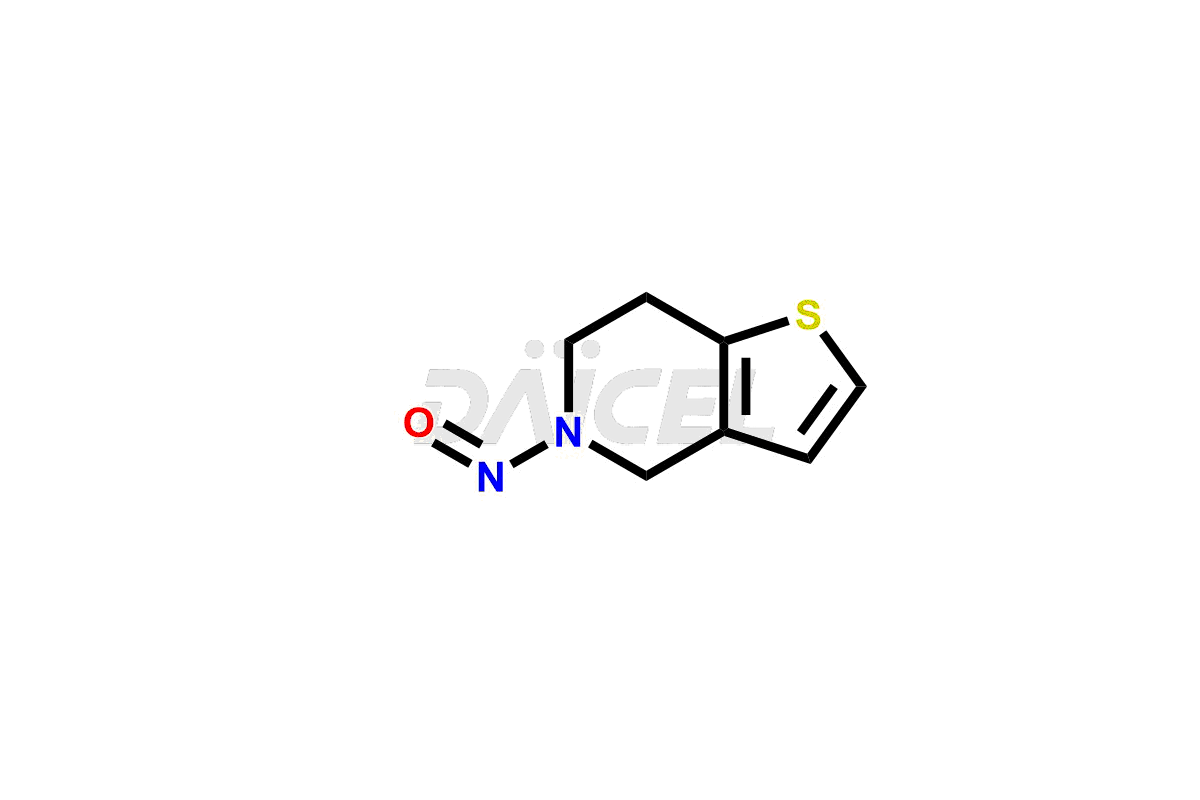
Pharma Standards


Accelerate Your Drug Development:
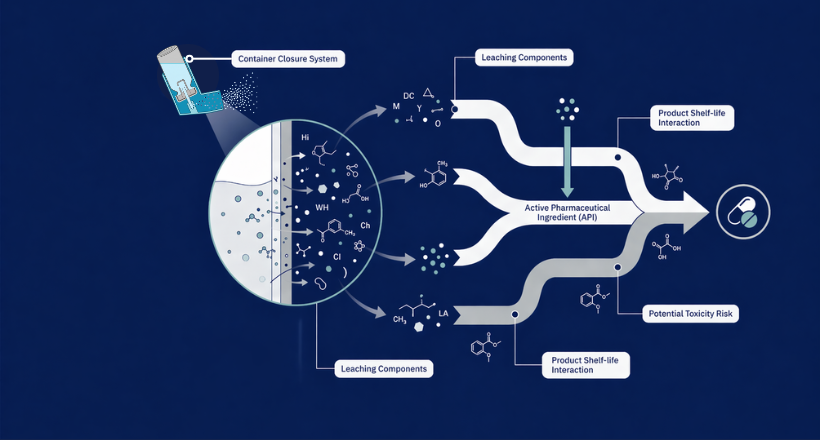
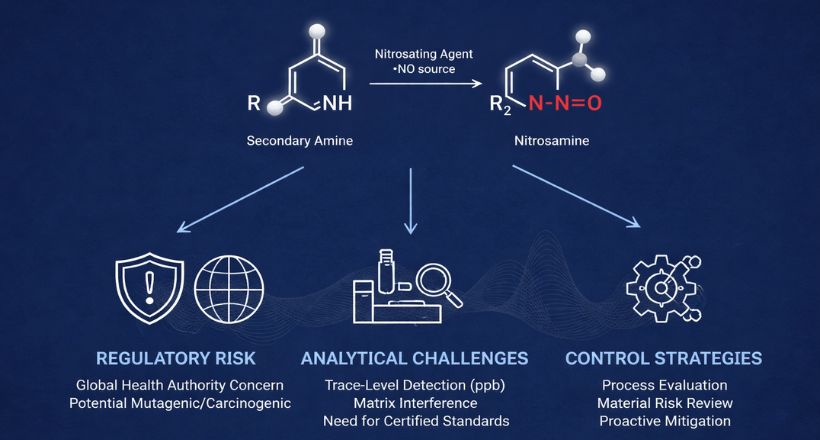
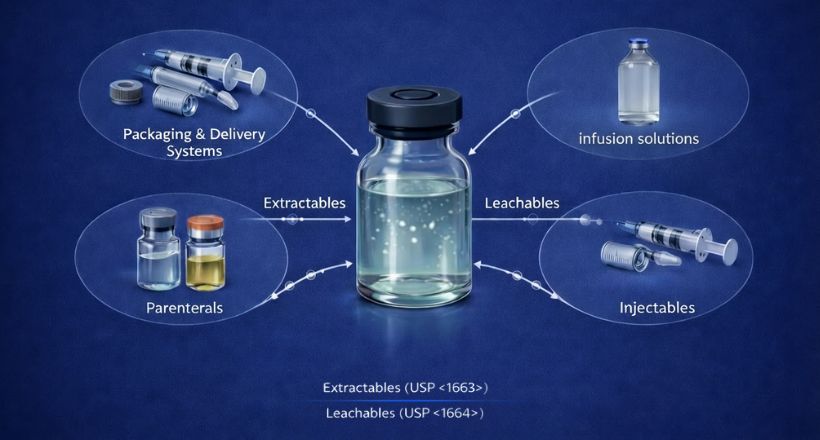
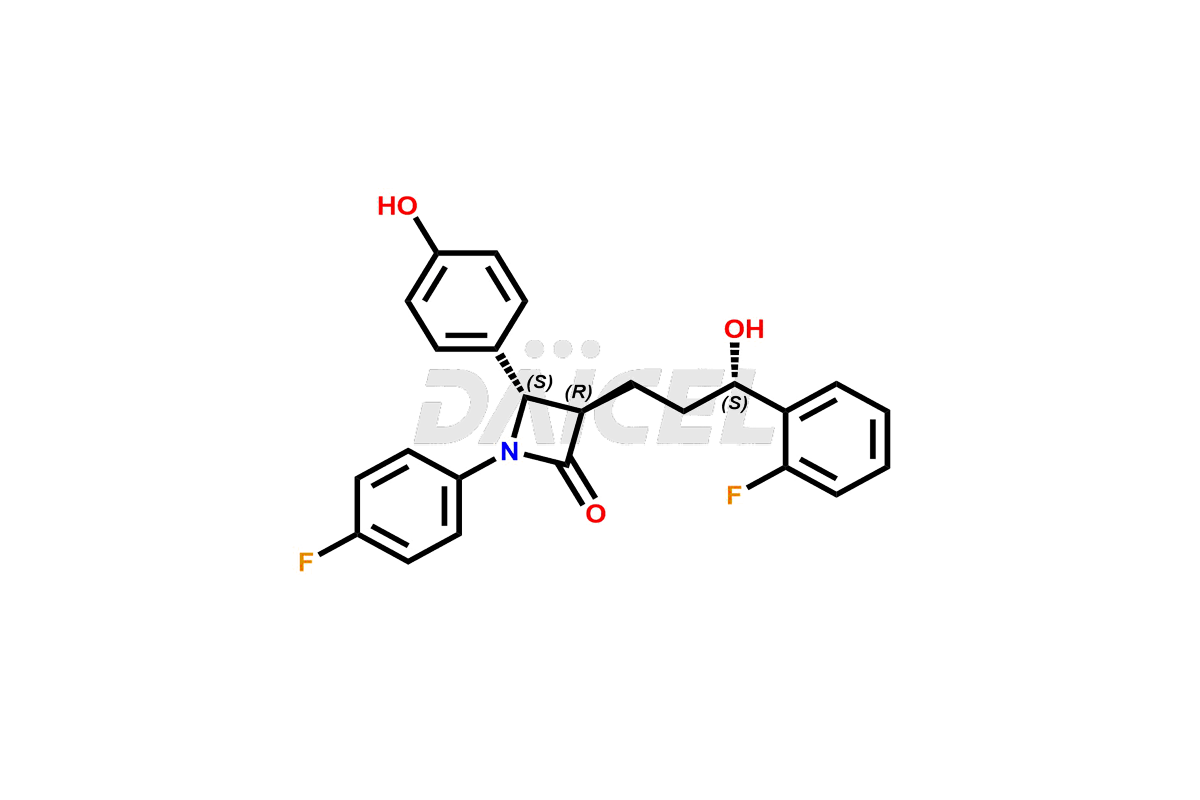
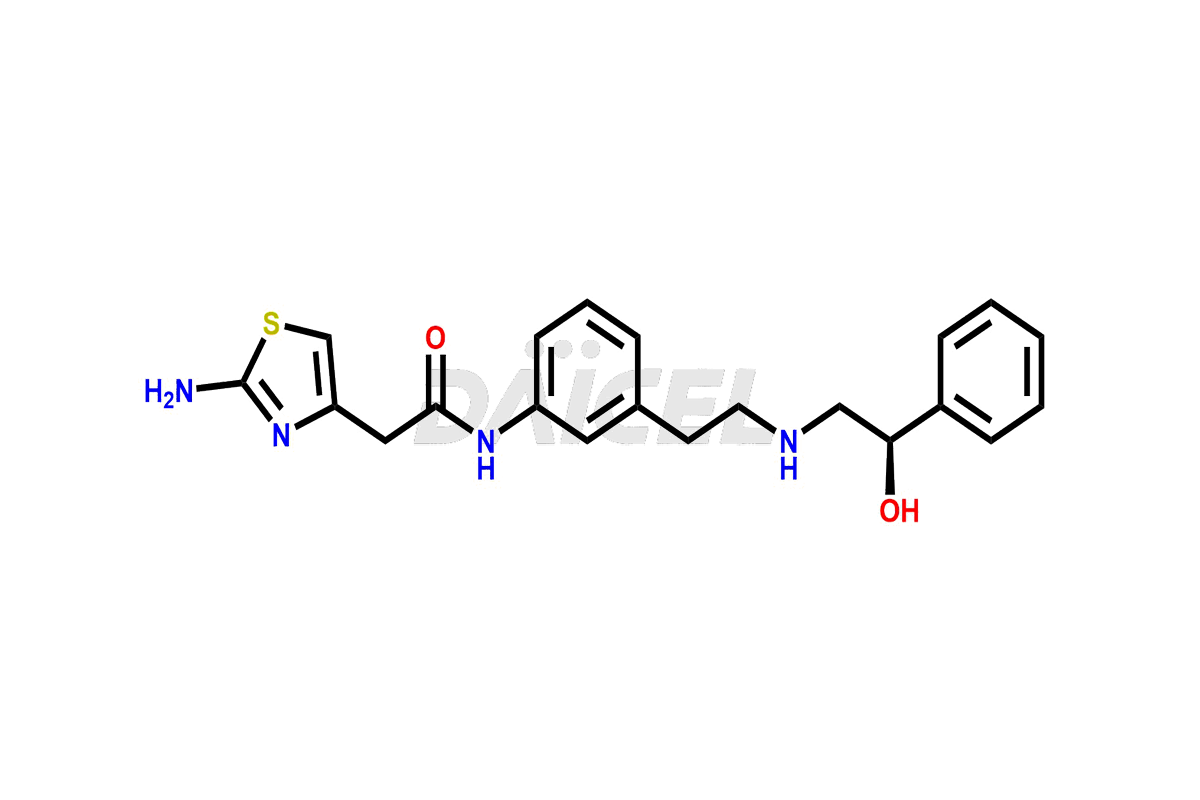
Our high quality certified impurity standards help you confidently meet regulatory expectations, and ensure product safety and efficacy.
Custom Synthesis


Fast Track Tailored Synthesis:
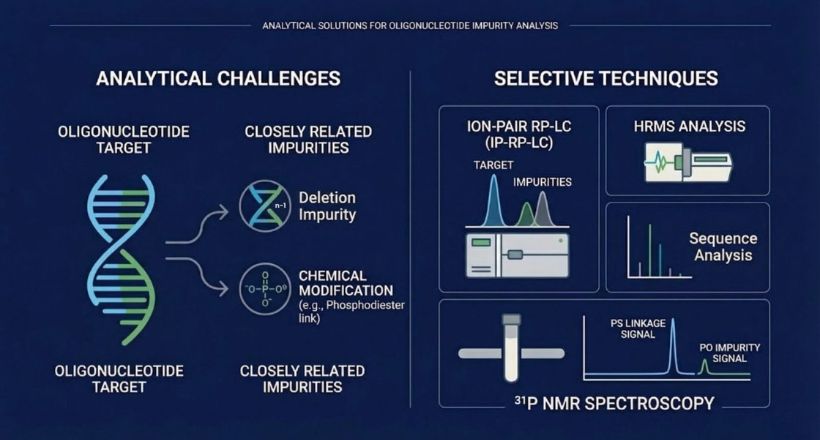
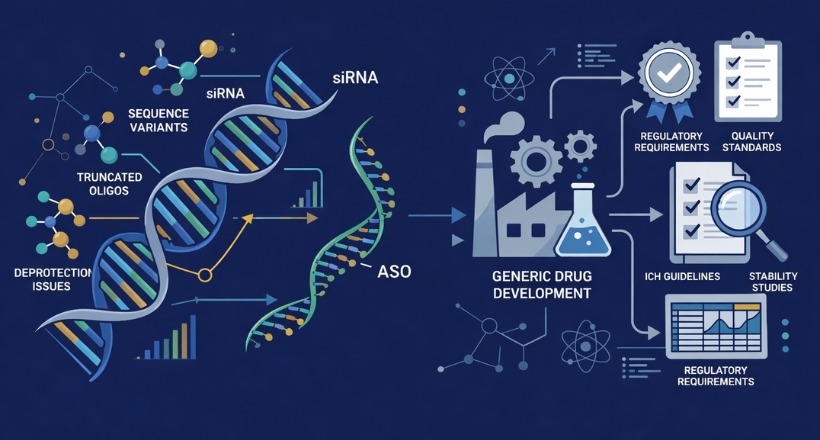
From milligrams to multi-grams, we provide tailored peptides, oligonucleotides, impurities, and isotope-labeled APIs, including custom peptide libraries designed to fast-track your discovery and drug development.
Pharma Services


Comprehensive Pharma Services:
A full suite of synthesis, purification, and analytical services to support your regulatory filings and accelerate drug development.
Daicel Pharma Standards
As a global leader in drug impurity standards, drug metabolites, and stable isotope-labelled compounds, we drive innovation in pharmaceutical development.
From synthesis to supply, we support every stage of the product development cycle, ensuring access to high-quality impurities and labelled compounds. Leveraging our in-house expertise, we’ve built an extensive catalogue spanning everything from small molecules to complex peptides, tailored to meet diverse research and quality control needs.


Our Facilities and Workflow