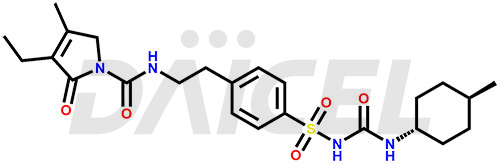
Glimepiride
General Information
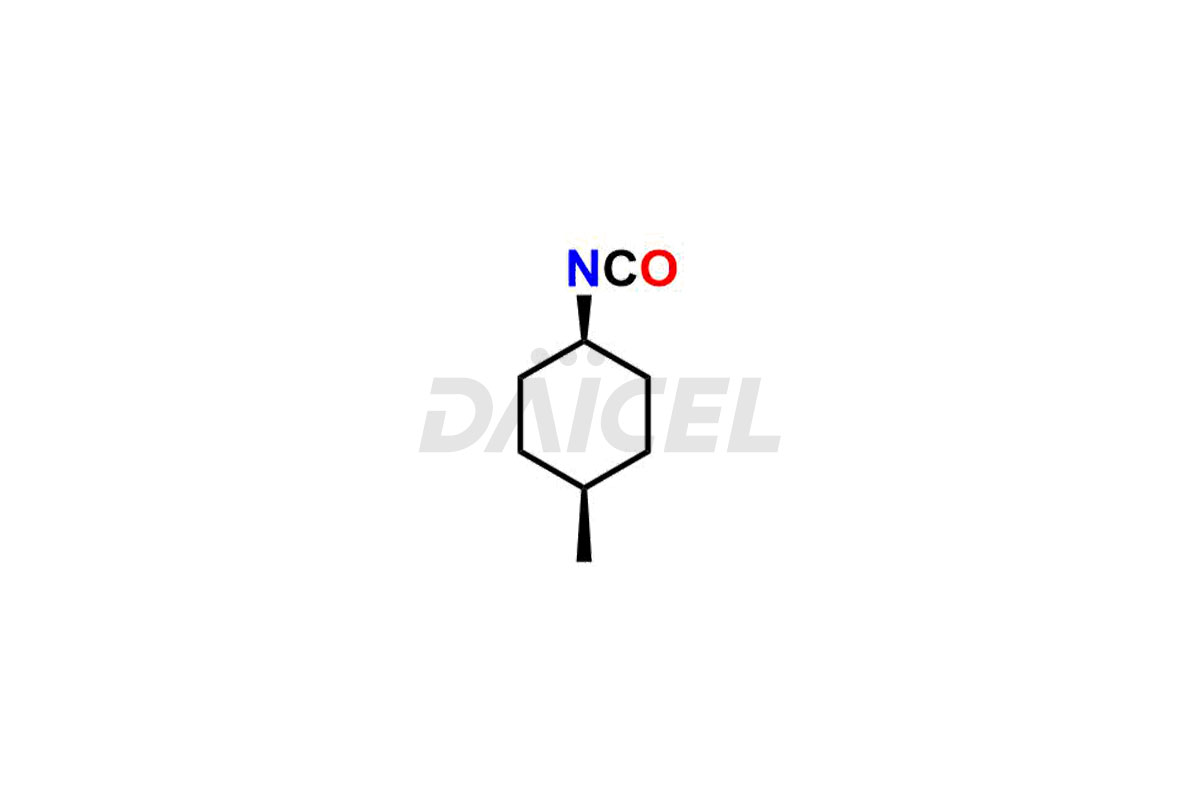
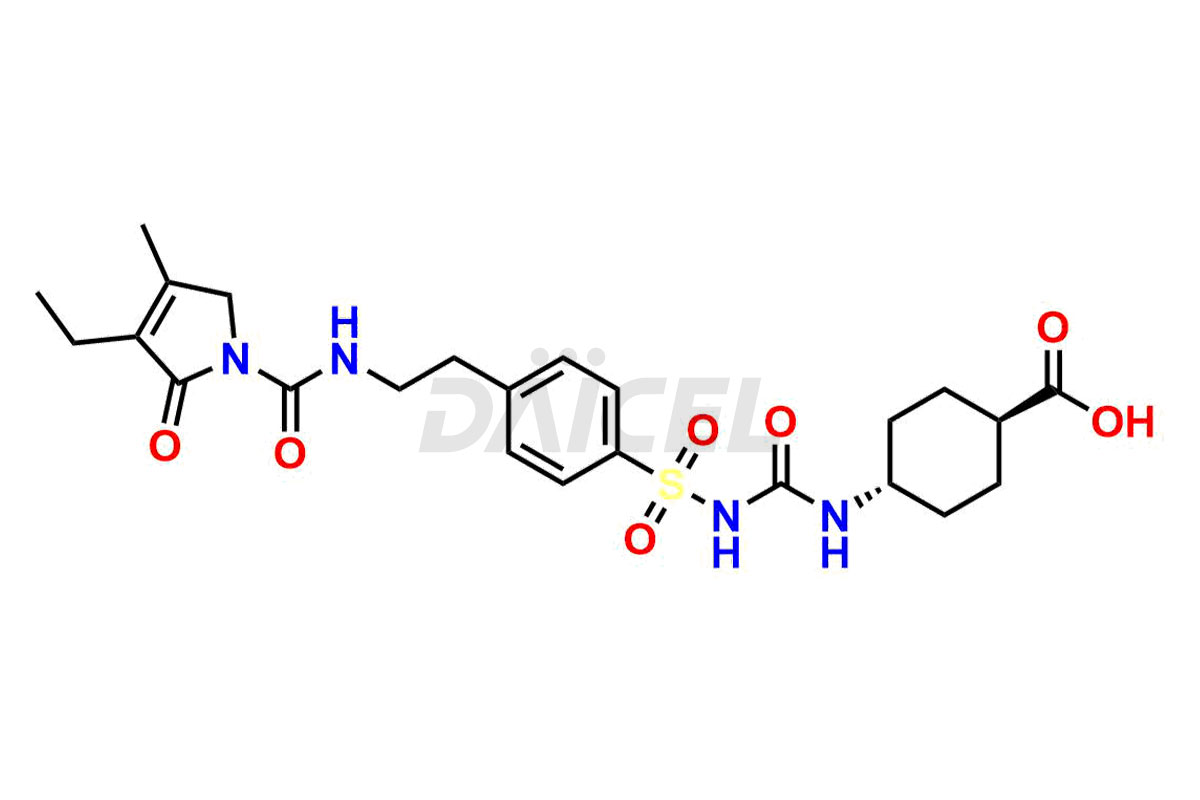
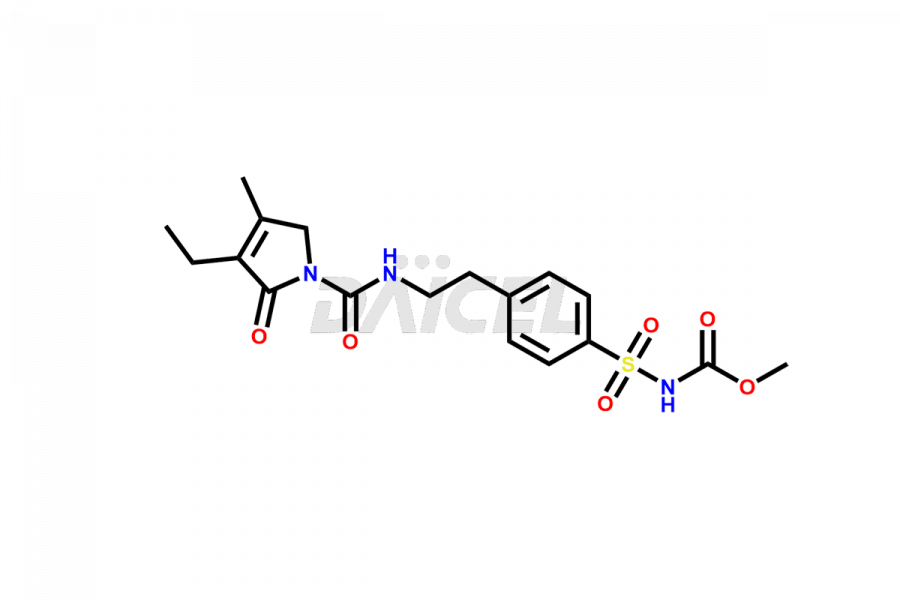
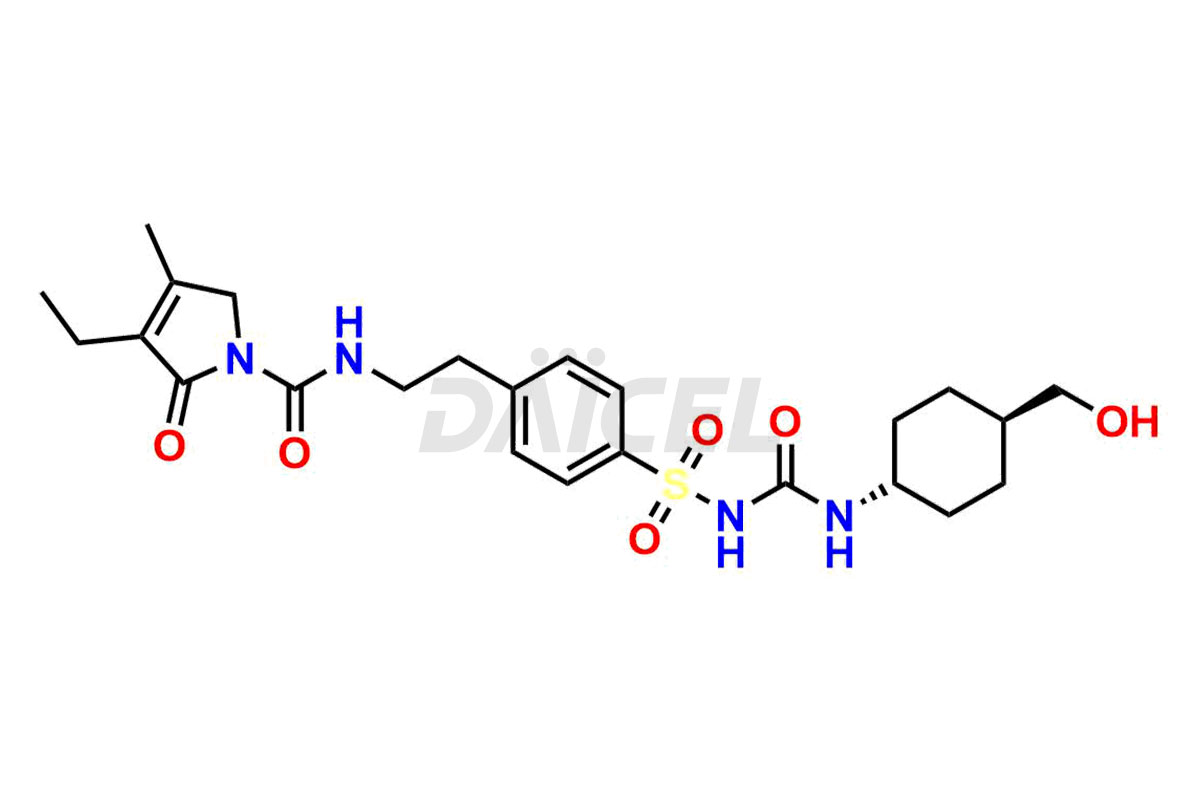
Glimepiride Impurities and Glimepiride
Daicel Pharma offers worldwide delivery options for a custom synthesis of Glimepiride impurities, including impurities such as Cis Isomer of MCI-Glimepiride, Cyclohexyl carboxyl Glimepiride, and Trans-hydroxy Glimepiride. These impurities play a vital role in evaluating the purity and safety of Glimepiride, an active pharmaceutical ingredient.
Glimepiride [CAS: 93479-97-1] is a long-acting third-generation sulfonylurea medication. It lowers blood sugar levels and stimulates the secretion of insulin. With its extended duration of action and hypoglycemic effects, Glimepiride is an effective option for managing diabetes.
Glimepiride: Use and Commercial Availability
Approved by the FDA for treating type 2 diabetes mellitus, Glimepiride is a second-generation sulfonylurea medication. It enhances glycemic control in adults. Glimepiride is used in cases where metformin is not tolerated. Amaryl is the brand name under which Glimepiride is available.
Glimepiride Structure and Mechanism of Action 
The chemical name of Glimepiride is 3-ethyl-4-methyl-N-(4-(N-((4-methylcyclohexyl)carbamoyl)sulfamoyl)phenethyl)-2-oxo-2,5-dihydro-1H-pyrrole-1-carboxamide. Its chemical formula is C24H34N4O5S, and its molecular weight is approximately 490.6 g/mol.
Glimepiride stimulates the release of insulin from functioning pancreatic cells.
Glimepiride Impurities and Synthesis
Glimepiride impurities can form during synthesis1, storage, or usage, resulting from various factors such as side reactions and degradation. Analytical methods like HPLC, LC-MS, NMR, and IR help analyze and quantify these impurities. Control measures include optimizing synthetic conditions, purification steps, and implementing advanced analytical technology. Proper storage conditions and packaging are essential to minimize impurity formation during storage. Stability studies help assess impurity profiles over time. Compliance with regulatory guidelines ensures the quality and safety of Glimepiride.
Daicel Pharma strictly adheres to cGMP standards and operates an analytical facility for synthesizing Glimepiride impurity standards. We provide a range of Glimepiride impurities, such as Cis Isomer of MCI-Glimepiride, Cyclohexyl carboxyl glimepiride, and Trans-hydroxy Glimepiride. Our impurity standards have a detailed Certificate of Analysis (CoA) and a comprehensive characterization report. The CoA encompasses data obtained through techniques, including 1H NMR, 13C NMR, IR, MASS, and HPLC purity analysis2. We give additional data, such as 13C-DEPT, on request. We can synthesize unknown Glimepiride impurities or degradation products. Each delivery has a comprehensive characterization report.
References
FAQ's
References
- Thennati, Rajamannar; Rehani, Rajeev Budhdev; Soni, Rohit Ravikant, Process for the preparation of trans-3-ethyl-2,5-dihydro-4-methyl-N-[2-[4-[[[[(4-methyl cyclohexyl) amino]carbonyl]amino]sulfonyl] phenyl]ethyl]-2-oxo-1H-pyrrole-1-carboxamide, Sun Pharmaceutical Industries Limited, India, US7132444B2, November 7, 2006
- Lehr, K. H.; Damm, P., Simultaneous determination of the sulfonylurea glimepiride and its metabolites in human serum and urine by high-performance liquid chromatography after pre-column derivatization, Journal of Chromatography, Biomedical Applications, Volume: 526, Issue: 2, Pages: 497-505, 1990
Frequently Asked Questions
Are Glimepiride impurities monitored over time?
Yes, impurities in Glimepiride are routinely monitored during stability studies to assess the long-term quality and safety of the drug.
How are the Glimepiride impurities identified?
Impurities in Glimepiride are identified by comparing their chromatographic or spectral characteristics with those of known impurities or reference standards.
Can Glimepiride impurities be removed or reduced?
Manufacturers strive to minimize impurities during synthesis, and purification techniques can be employed to reduce impurity levels.
How should Glimepiride impurities be stored in terms of temperature?
The recommendation is to store Glimepiride impurities at a controlled room temperature, within 2-8 °C.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.