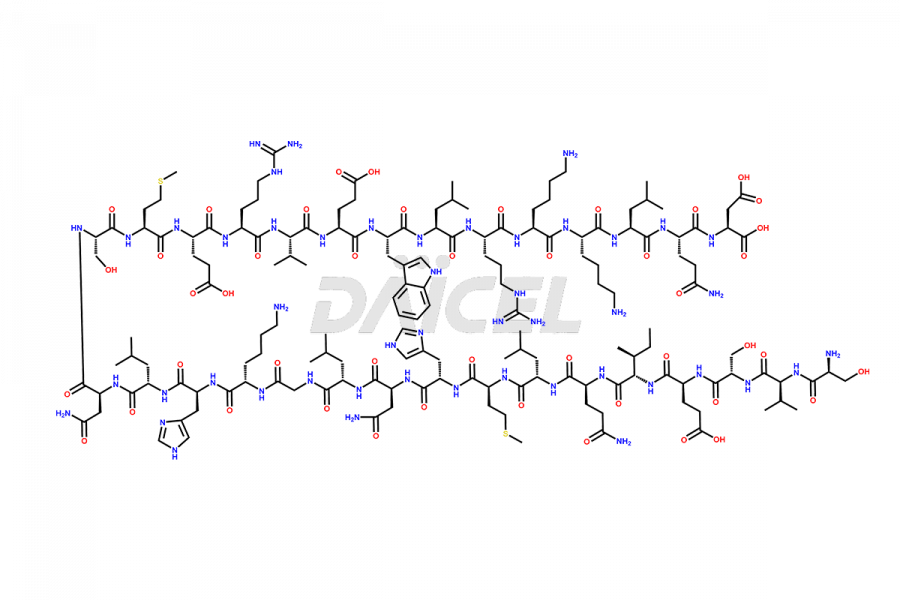
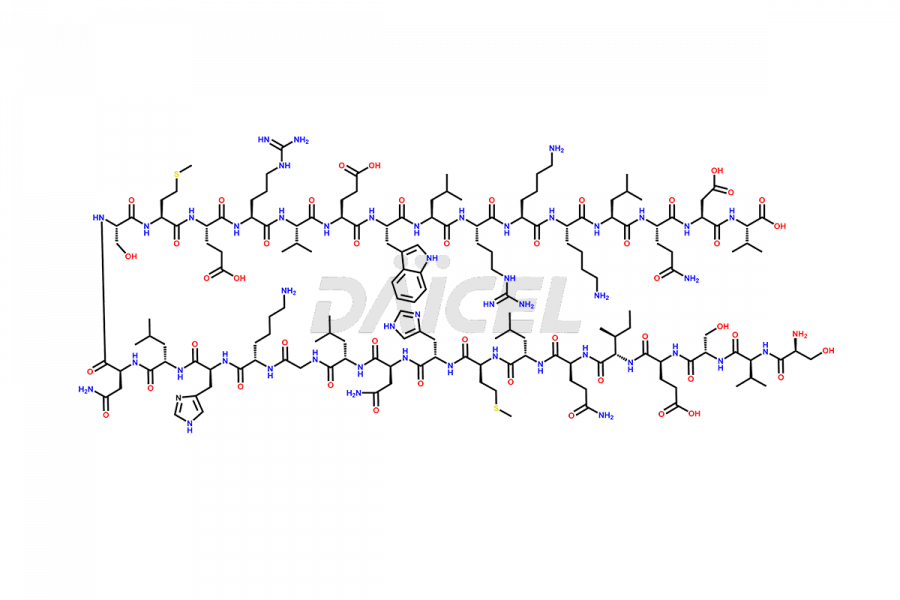
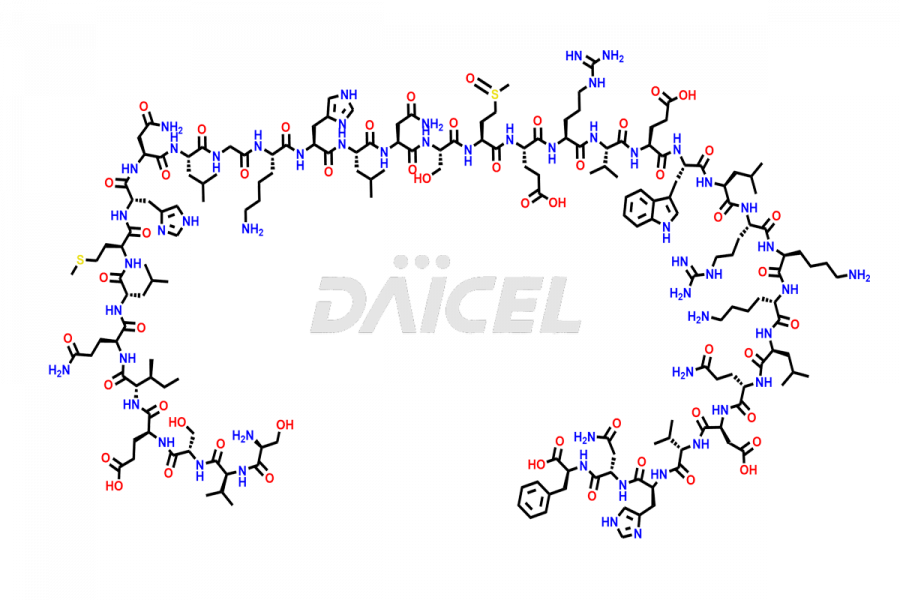
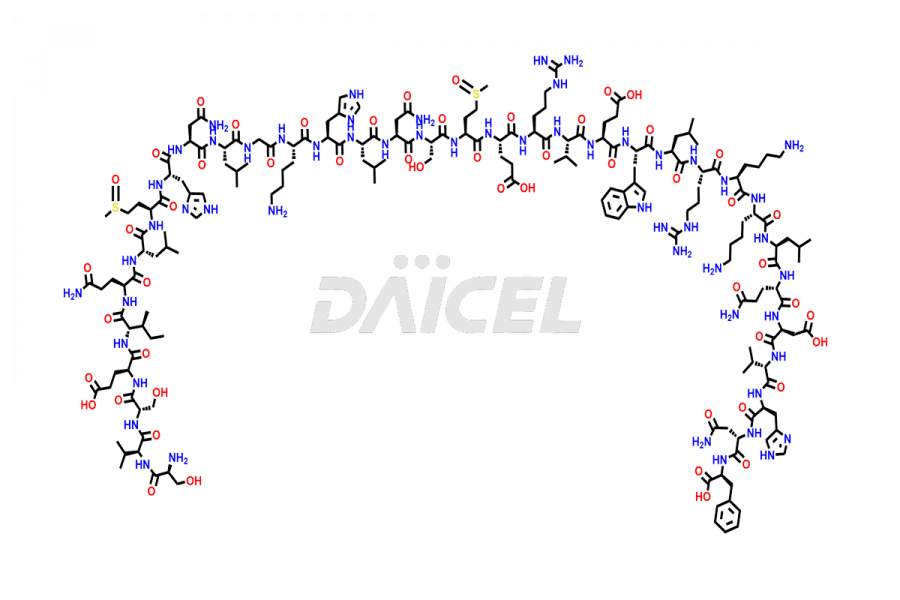
Teriparatide
General Information
Teriparatide Impurities and Teriparatide
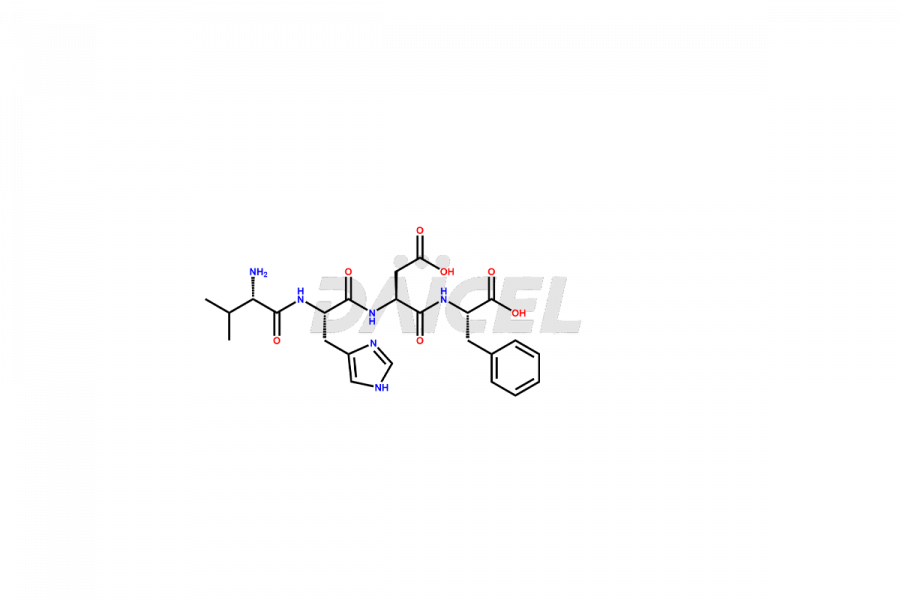
Daicel Pharma produces high-quality Teriparatide impurities, such as [Met(O)-8,18]-Teriparatide and Dimer equivalent, that can assist in the analysis of the quality, stability, and biological safety of the Teriparatide active pharmaceutical ingredient. In addition, we provide custom synthesis of Teriparatide impurities and can deliver them worldwide.
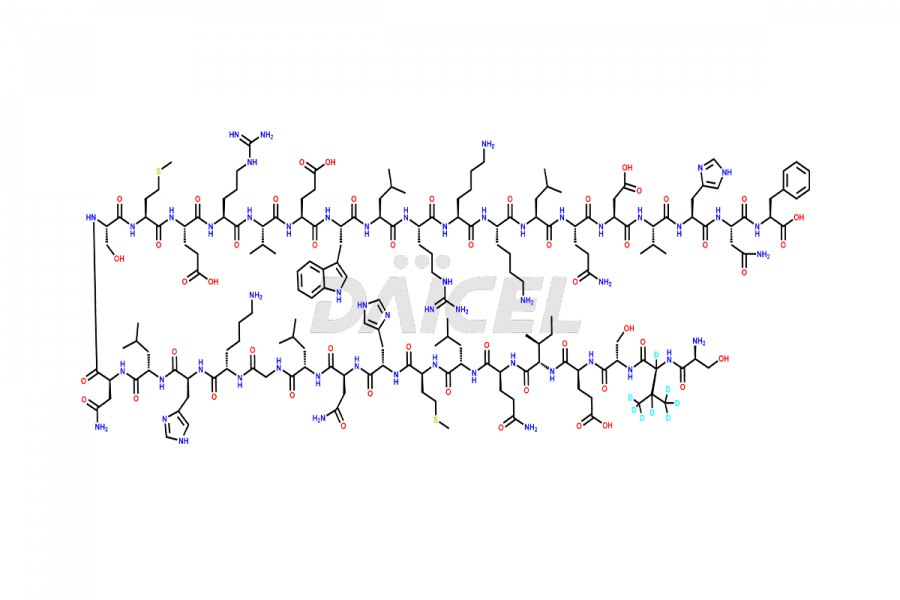
Teriparatide [CAS: 52232-67-4] is a recombinant human parathyroid hormone analog (PTH 1-34). It has an identical sequence of 34 N-terminal amino acids of the 84-amino acid human parathyroid hormone. It is primarily responsible for promoting bone growth, increasing bone density, and reducing bone loss. Studies have demonstrated that Teriparatide increases bone mass and density and prevents fractures in individuals with osteoporosis.
Teriparatide: Use and Commercial Availability
Teriparatide is for the treatment of osteoporosis in postmenopausal women and men with an increased risk of fracture. It acts on osteoblasts (bone-forming cells) and stimulates bone formation. In addition, it’s for use in women and men with glucocorticoid-induced osteoporosis at high risk of fracture. Teriparatide is the only anabolic medicine approved for the treatment of osteoporosis. It is sold under the brand names- Forteo, Bonsity, Sondelbay, and Parathar. Teriparatide is administered through a glass cartridge, pre-assembled into a delivery device (pen) via subcutaneous injection into the abdominal wall or anterior thigh. It can lead to a 13% increase in bone mass over two years of therapy when given intermittently.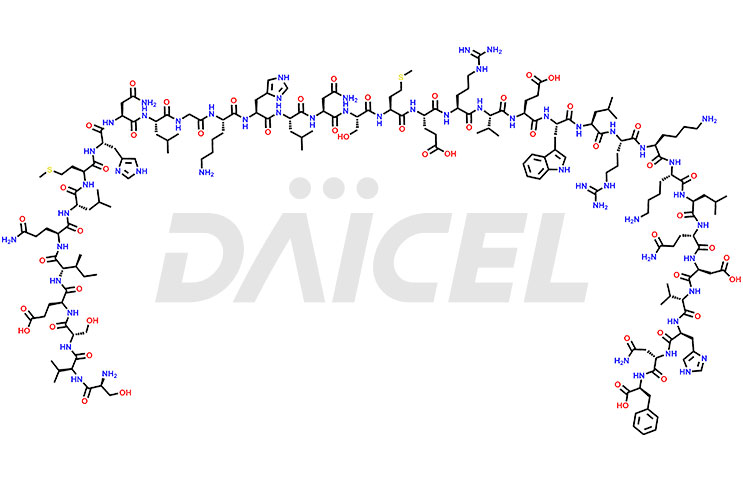

Teriparatide Structure and Mechanism of Action
The chemical formula of Teriparatide is C181H291N55O51S2, and its molecular weight is approximately 4117.8 g/mol.
The endogenous 84-amino-acid parathyroid hormone (PTH) is the primary regulator of calcium and phosphate metabolism in bone and kidney. The physiological actions of PTH include regulation of bone metabolism, intestinal calcium absorption, and renal tubular reabsorption of calcium and phosphate. Teriparatide is a synthetic form of parathyroid hormone (PTH) that binds to PTH/PTHrP receptors and activates downstream osteoblast signaling, which leads to enhanced bone formation and increased bone density in osteoporosis patients.
Teriparatide Impurities and Synthesis
During the synthesis1 and storage of Teriparatide, several impurities are produced like Trpp23-teriparatide oxidation impurities, including Trp23(C=O)-Teriparatide, Trp23(OH)-Teriparatide, Met8(O)-Teriparatide, Met18(O)-Teriparatide, Met8,18(O)-Teriparatide, and more2. Controlling the number of impurities in the Teriparatide drug helps to maintain its safety and efficacy.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for Teriparatide impurity standards, including [Met(O)-8,18]-Teriparatide and Dimer equivalent. The CoA includes complete characterization data3, such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity, and we provide 13C-DEPT and CHN on request. We also give a complete characterization report on delivery. Daicel has the technology and expertise to prepare any unknown Teriparatide impurity or degradation product. We also provide labeled compounds to quantify the efficacy of generic Teriparatide. Daicel offers highly pure isotope-labeled standards4 of Teriparatide for bioanalytical research and BA/BE studies with isotope data in CoA.
References
FAQ's
References
- Tregear, Geoffrey W.; Van Rietschoten, Jurphaas; Greene, Elizabeth; Niall, Hugh D.; Keutmann, Henry T.; Parsons, John A.; O'Riordan, Jeffrey L. H.; Potts, John T. Jr., “Solid-phase synthesis of the biologically active N-terminal 1-34 peptide of human parathyroid hormone”, Hoppe-Seyler's Zeitschrift fuer Physiologische Chemie, Volume: 355, Issue: 4, Pages: 415-21,1974 DOI
- WANG, Wei, JIANG,Xuban, YIN, Chuanlong, ΤΑΟ, Anjin, YU,Pinxiang, Hybio Pharmaceutical Co., Ltd, “Teriparatide Impurity F” PCT application, WO2021114788A1, June 17, 2021
- Kothari, Ruchi; Kumar, Vinod; Jena, Rajender; Tunga, Rashbehari; Tunga, Binita Shrivastava, “Modes of degradation and impurity characterization in rhPTH (1-34) during stability studies”, PDA Journal of Pharmaceutical Science and Technology, Volume: 65, Issue: 4, Pages
- Chansel, D.; Sraer, J.; Morgat, J. L.; Hesch, R. D.; Ardaillou, R., “Preparation of biologically active tritium-labeled 1-34 human parathyroid hormone”, FEBS Letters, Volume: 78, Issue: 2, Pages: 237-43, 1977
Frequently Asked Questions
How are Teriparatide impurities synthesized?
The synthesis of Teriparatide impurities involves a variety of chemical reactions, such as oxidation, reduction, hydrolysis, and esterification. The synthetic pathways will depend on the study of the specific impurities.
What are the analytical techniques used to identify Teriparatide impurities?
Analytical techniques such as high-performance liquid chromatography (HPLC), and liquid chromatography mass spectrometry (LC-MS) help to identify Teriparatide impurities.
How are Teriparatide impurities removed from the drug?
Teriparatide impurities can be removed from the drug using various purification techniques such as chromatography, filtration, and extraction. These techniques help to isolate and remove impurities from Teriparatide.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.